Four failure points.
All modeled before the trial begins.
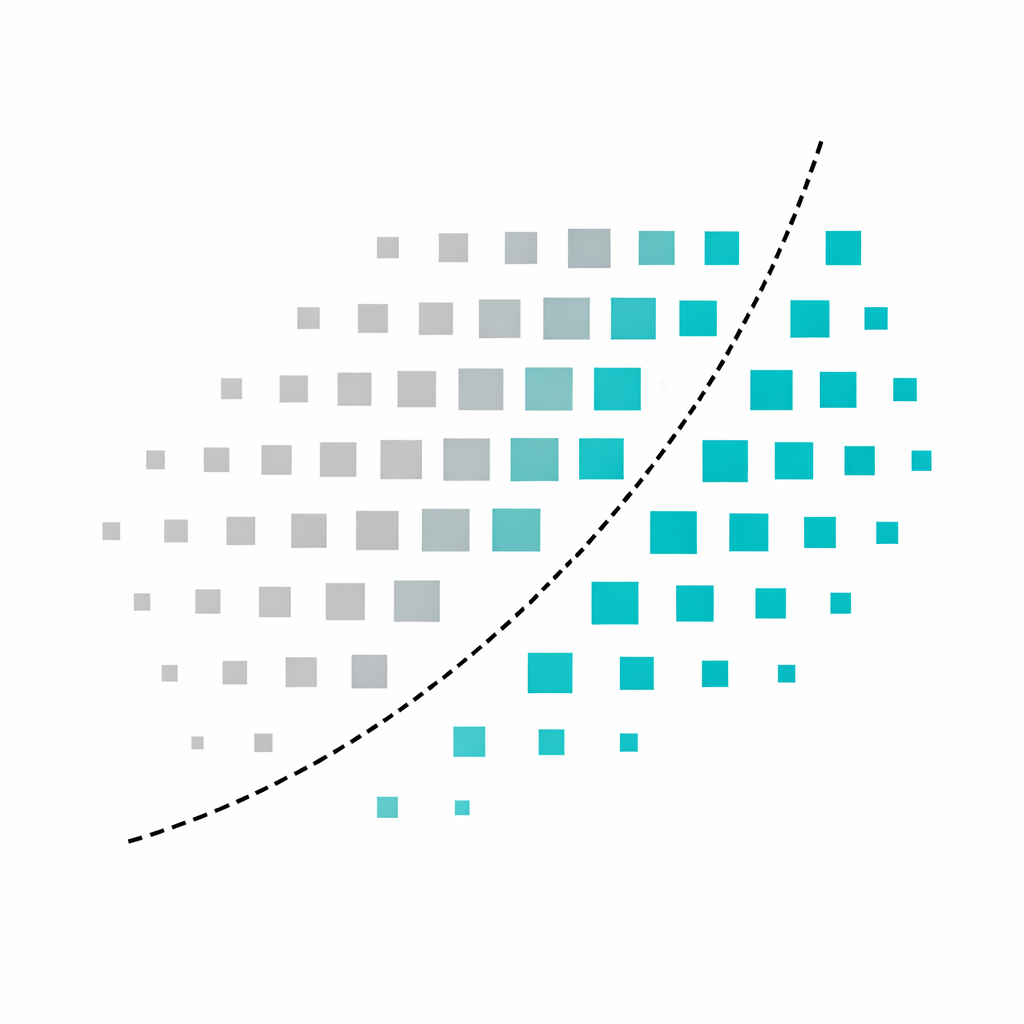
Identifiable from behavioral profile before randomization.
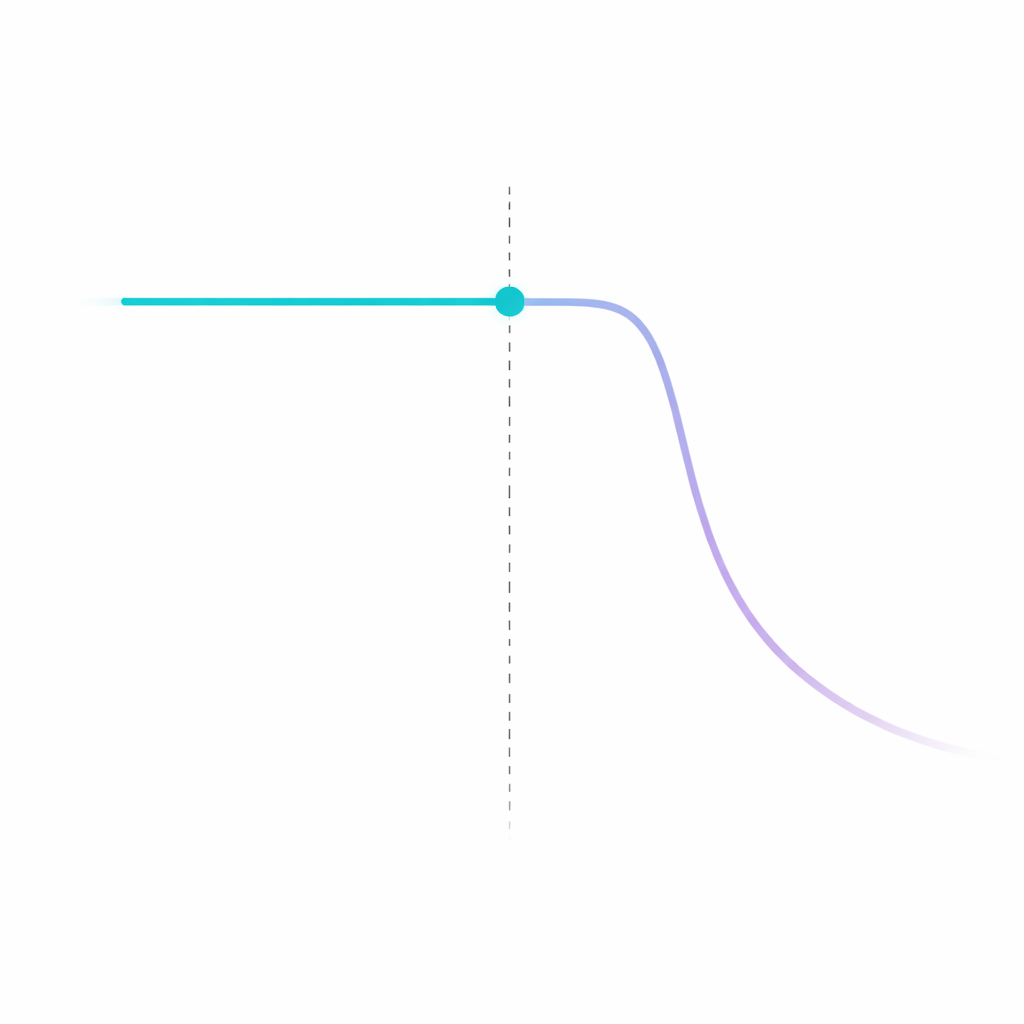
Detectable at baseline. Flagged before the window closes.

Predicted before it is observable. Intervened before it is irreversible.
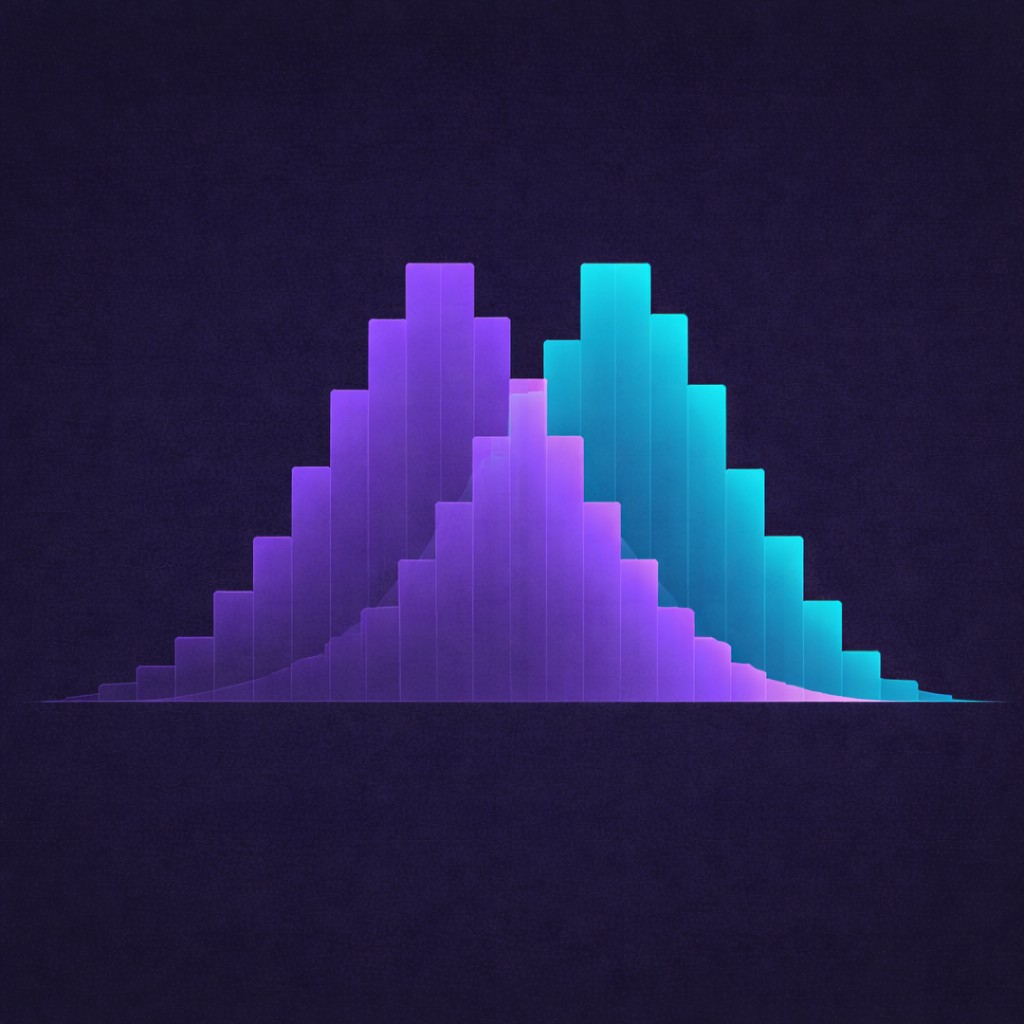
Population contamination risk surfaced before the data is dirty.